|
What is the dilution factor, i.e, how many more times dilute is it than the original concentration?įinal Volume = Volume of Diluent (Distilled Water here) + 100 (Volume of Stock ) = 800 mLĭilution Factor = Final Volume / Volume of Stock Aliquotĭilution Factor is 8 or the stock is 1/8 times diluted. You do a dilution by combining 100 ml volume of NaCl plus 700 ml unit volumes of Distilled water. Serial Dilution Calculation: Problems and Solutions Tube 4 (Dilution 4) - Take 900uL of sterial water in tube 4 and add 100uL from Tube 3 - so tube 4 will have10^2 cells/mL or 100 cells/mL. Tube 3 (Dilution 3) - Take 900uL of sterial water in tube 3 and add 100uL from Tube 2 - so tube 3 will have10^3 cells/mL. Tube 2 (Dilution 2) - Take 900uL of sterial water in tube 2 and add 100uL from Tube 1 - so tube 2 will have10^4 cells/mL. Tube 1 (Dilution 1) - Take 900uL of sterial water in tube 1 and add 100uL of bacterial stock - so tube 1 will have10^5 cells/mL. Stock of Bacterial Culture - 10^6 Bacterial Cells You need to do Four 10 fold dilution to a volume of 1mL to get 10^2 cells/mL final concentration. You have a bacterial culture, which has 10^6 bacterial cells and you want 100 cells/mL how to dilute the culture to get 100cells/mLįinal Concentration Required - 100 cells/mL or 10^2 cells/mL Serially dilute the bacterial culture, Bacterial culture is generally serially diluted for plating (for bacterial isolation), for re-inoculation or for other applications like checking optical density, etc. How to do Serial Dilution of Bacterial Culture?ĭilution of bacterial culture can be performed the same way as explained above. Stock solution is 1 M, after 1000 fold dilution it becomes - 1/1000 = 0.001 M. Label the tube as Dilution 1.įrom Dilution 1 tube take 10mL and mix 90mL of diluent and Label it as Dilution 2.įrom From Dilution 2 tube take 10mL and mix 90mL of diluent and Label it as Dilution 3.Īs we have done 10 fold dilutions, the tube 3 (Dilution 3) will be 1000 fold diluted than the stock. Mix 10mL of 1M stock with 90mL of Diluent. To do a 10 fold dilution of 1M Stock solution to 100mL, Since we need to do 10 fold dilution, dilution factor is 10.ĭiluent Volume required = 100mL - 10mL = 90mL. Lets see how to serially dilute 1 M stock solution to a final volume of 100 mL.ĭilution Factor = Final Volume / Volume of Stock. 10 fold dilution is actually 10^-1 dilution (Inverse of Dilution Factor: 1/10).ĭiluting a 1 M (Molar) Stock solution 1000 fold by serial Dilution.ġ000 fold serial dilution can be done by three 10 fold dilutions. Dilution is the inverse of Dilution Factor. The ratio of the Final volume to the aliqout volume of the stock is known as Dilution Factor (DF). It does not store any personal data.To dilute a stock solution, Mix the stock solution with diluent. 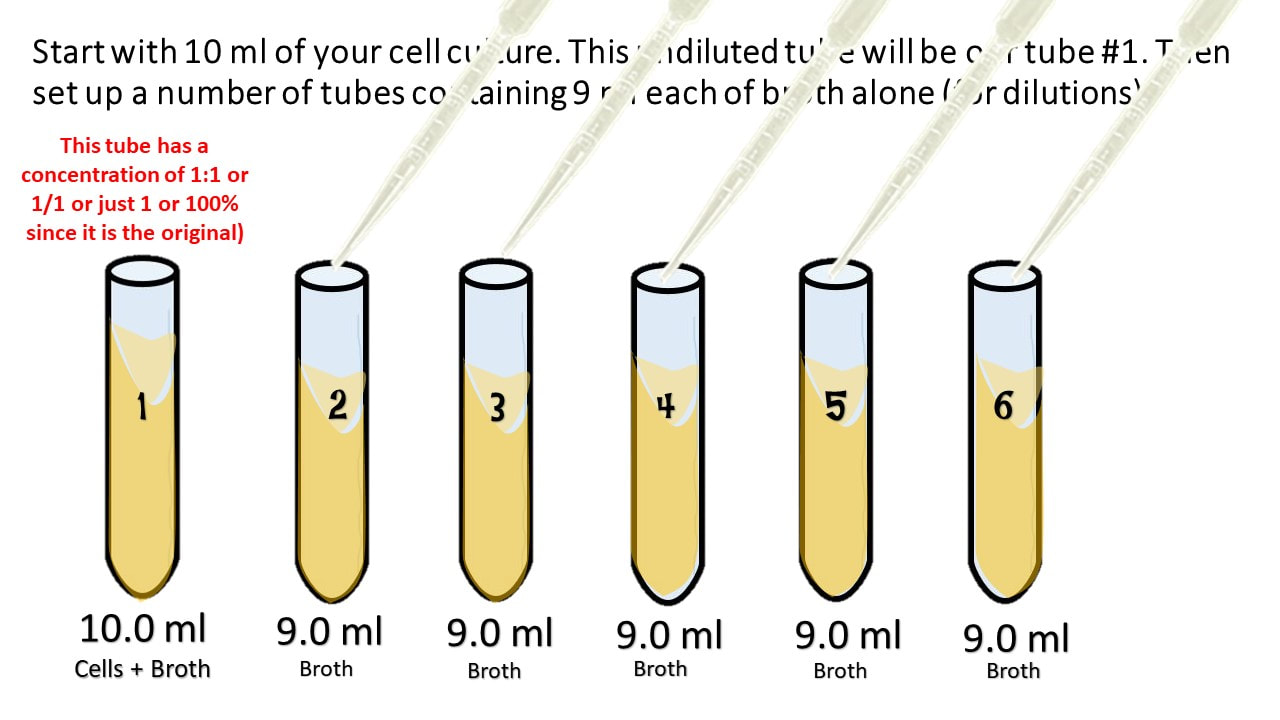
The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. The cookie is used to store the user consent for the cookies in the category "Performance". This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is used to store the user consent for the cookies in the category "Other.  
The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". 
These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
